 The Annual Report to the Nation on the Status of Cancer, 1975–2009, shows that overall cancer death rates continued to decline in the United States among both men and women, among all major racial and ethnic groups, and for all of the most common cancer sites, including lung, colon and rectum, female breast, and prostate. However, the report also shows that death rates continued to increase during the latest time period (2000 through 2009) for melanoma of the skin (among men only) and for cancers of the liver, pancreas, and uterus. The special feature section on human papillomavirus (HPV)-associated cancers shows that incidence rates are increasing for HPV-associated oropharyngeal and anal cancers and that vaccination coverage levels in the U.S. during 2008 and 2010 remained low among adolescent girls.
The Annual Report to the Nation on the Status of Cancer, 1975–2009, shows that overall cancer death rates continued to decline in the United States among both men and women, among all major racial and ethnic groups, and for all of the most common cancer sites, including lung, colon and rectum, female breast, and prostate. However, the report also shows that death rates continued to increase during the latest time period (2000 through 2009) for melanoma of the skin (among men only) and for cancers of the liver, pancreas, and uterus. The special feature section on human papillomavirus (HPV)-associated cancers shows that incidence rates are increasing for HPV-associated oropharyngeal and anal cancers and that vaccination coverage levels in the U.S. during 2008 and 2010 remained low among adolescent girls.
The report, produced since 1998, is co-authored by researchers from the American Cancer Society (ACS), the Centers for Disease Control and Prevention (CDC), the National Cancer Institute (NCI), and the North American Association of Central Cancer Registries (NAACCR). It appears early online in the Journal of the National Cancer Institute and will be published in print issue 3, volume 105.
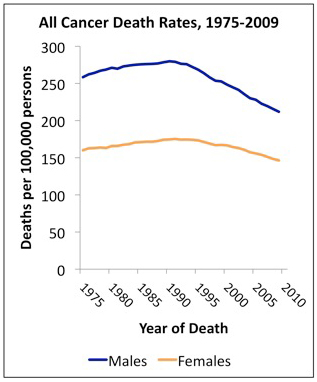
The decline in overall cancer death rates continues a trend that began in the early 1990s. From 2000 through 2009, cancer death rates decreased by 1.8 percent per year among men and by 1.4 percent per year among women. Death rates among children up to 14 years of age also continued to decrease by 1.8 percent per year. During 2000 through 2009, death rates among men decreased for 10 of the 17 most common cancers (lung, prostate, colon and rectum, leukemia, non-Hodgkin lymphoma, kidney, stomach, myeloma, oral cavity and pharynx, and larynx) and increased for melanoma of the skin and cancers of the pancreas and liver. During the same 10-year period, death rates among women decreased for 15 of the 18 most common cancers (lung, breast, colon and rectum, ovary, leukemia, non-Hodgkin lymphoma, brain and other nervous system, myeloma, kidney, stomach, cervix, bladder, esophagus, oral cavity and pharynx, and gallbladder) and increased for cancers of the pancreas, liver, and uterus.